|
Rather than relying on passive diffusion, the pumps can move molecules such as ions up an electrochemical gradient because they can utilize energy generated, for example, from hydrolysis of ATP ( Pedersen & Carafoli 1987a Pedersen & Carafoli 1987b). The transporters that are perhaps furthest removed conceptually from the channels are the primary active transporters, some of which are also referred to as “pumps”. Other types of membrane proteins can also mediate movement of ions or other moieties across cell membranes but do not fall into the category of ion channels, and are referred to as “transporters”. Another class of channels that primarily provides a conduit for water across the plasma membrane are the water channels, or aquaporins ( Morelle et al. Conversely, some ion channels exist in non-excitable cells where their role is to ensure ion and fluid homeostasis or supply the necessary ingredients or conditions for biological functions such as hormone synthesis or mucous secretion. This property, together with their often remarkable ion selectivity (K + channels can readily permit K + movement but are proficient at limiting Na + leak, which bears similar charge to K + but is slightly smaller), has endowed cells with the ability to communicate on the millisecond timescale using action potentials. Thus, ion channels rely on an electrochemical gradient for their activity. Ion channels facilitate passive yet often rapid (e.g., tens of thousands of ions per second per channel pore) movement of aqueous ions across the otherwise forbidding barrier of the hydrophobic plasma membrane that evolved at least partly to prevent such leakage. Ion channels constitute a numerous and eclectic class of membrane proteins that are essential for the function of most if not all cell types from the archaebacteria through to the plethora of specialized cells in higher mammals. We compare functional and/or physical interactions between the ubiquitous KCNQ1 potassium channel and various active transporters, and examine other newly discovered chansporter complexes that suggest we may be seeing the tip of the iceberg in a newly emerging signaling modality. 
Here, we review the first known members of this new class of macromolecular complexes that we term “chansporters”, explore their biological roles, and discuss the pathophysiological consequences of their disruption. More recently, evidence is emerging of direct physical interaction between true ion channels, and some primary or secondary active transporters. It is well known that proteins in each of these classes work in concert with members of the other classes to ensure, for example, ion homeostasis, ion secretion, and restoration of ion balance following action potentials. The primary active transporters (including H +/K +-ATPases and Na +/K +-ATPases) utilize ATP hydrolysis as an energy source to power uphill transport. The secondary active transporters (such as SLC superfamily solute transporters) achieve this by coupling uphill movement of the substrate to downhill movement of another ion, such as sodium. In contrast, active transporters can move moieties up their electrochemical gradient. 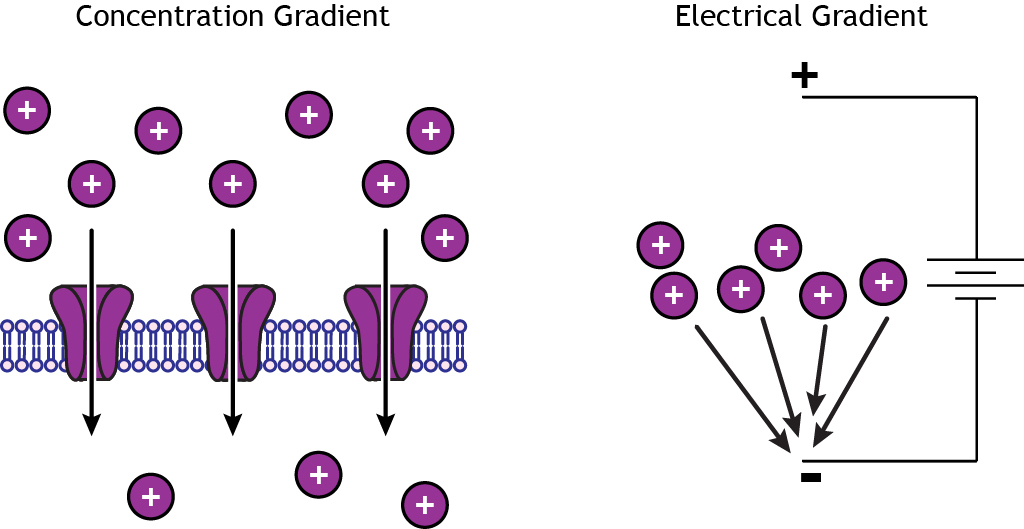
Ion channels provide a pore that permits often rapid, highly selective, and tightly regulated movement of ions down their electrochemical gradient. All living cells require membrane proteins that act as conduits for the regulated transport of ions, solutes and other small molecules across the cell membrane.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |



 RSS Feed
RSS Feed
